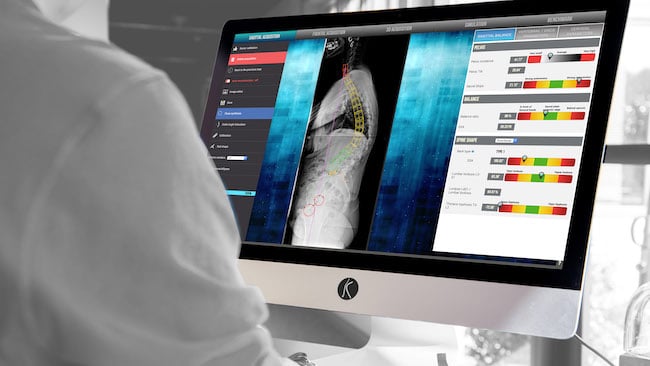
SMAIO (Software, Machines and Adaptative Implants in Orthopaedics – Euronext Growth Paris ISIN: FR0014005I80 / Ticker: ALSMA, eligible for PEA-PME equity savings plans), a French player specialized in complex spine surgery with a global offer comprising software, adaptive implants and related services, today announced that it has received FDA 510(k) clearance for its Balance Analyzer 3D surgery planning software.
Balance Analyzer 3D is spinal realignment planning software using medical imaging of the patient’s spine. Since receiving CE marking in 2014, it has been an integral part of the i-plan surgical planning solution marketed by SMAIO within its comprehensive i-kontrol platform that allows surgeons to treat spinal pathologies in a safe, effective and lasting way.
Philippe ROUSSOULY, CEO of SMAIO, stated: “This 510(k) clearance for the current version of our Balance Analyzer 3D planning software represents a major step in our American market penetration strategy that will be based, in the coming years, on our partnership with NuVasive. It lays the foundations for the co-developments the two companies have agreed to carry out over the next two years, and in the very near term will enable us both to initiate a certain number of technological and scientific collaborations with centers of excellence in the United States. This clearance illustrates the added value of SMAIO’s project that aims to develop, globally, the most cutting-edge solution for planning and performing spine surgery taking into account the specificities of each patient”.
Obtaining 510(k) clearance from the FDA is a key element of the partnership and licensing agreement signed in Q1 2022 between SMAIO and NuVasive, a global leader in spine technology innovation.
Source: SMAIO.