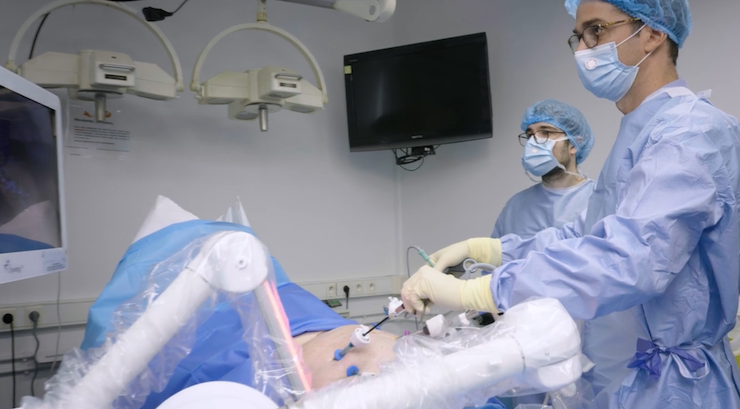
Moon Surgical has received the United States Food and Drug Administration (FDA) clearance for its unique Maestro surgical robotics system.
“At Moon Surgical, we are creating a new category of surgical robotics that will change the scale at which robotics can be deployed and enable healthcare providers to do more with the tools they already have,” said Anne Osdoit, CEO of Moon Surgical, and a Partner in Sofinnova Partners’ medtech accelerator, MD Start. “The clearance of Maestro by the FDA is a significant step forward on this path. It enables select high-volume surgical centers in the U.S. and the patients they serve to gain access to the capabilities offered by Maestro. It will also provide us with invaluable insights about its use in a real life setting.”
Maestro is designed to support surgeons and patients in the 18.8 million annual soft tissue surgical procedures not currently supported by telerobotic systems on the market. Maestro is small, adaptable, and can be integrated into existing clinical workflows. Its capabilities can bolster operating room efficiency and allow for alternative labor models.
“We have seen tremendous interest from surgical practices in the U.S. and are eager to see surgeons and their staff using Maestro for the benefit of their patients in that geography,” commented Professor Brice Gayet, a visionary laparoscopist and co-founder of Moon Surgical. “Maestro will dramatically change the way in which the standard of care is performed in soft tissue surgery, for millions of patients each year.”
Moon Surgical is currently finalizing the development of its first commercial system.
Source: Moon Surgical