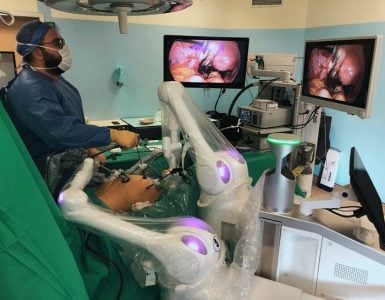
Prof. Ye and team celebrating the successful completion of first-in-man trial with Carina™ RAS Platform.
Ronovo Surgical has announced the successful completion of its first-in-man (FIM) trial with the Carina™ RAS Platform.
The world’s first human case performed with Carina™ was led by famed urology oncology expert, Prof. Dingwei Ye, and his team at the Fudan University Shanghai Cancer Center (FUSCC). A nationally recognized urology expert, Prof. Ye presides as Vice President and Chairman of Urology at FUSCC, and he also chairs the Urology committee of the Chinese Anti-Cancer Association.
For its debut in human patients, Carina™ was utilized to perform a radical prostatectomy on a 66-year-old patient who was diagnosed with prostate cancer and Gleason score: 4+4=8. The successful procedure saw the robotic system maneuver nimbly in deep and narrow pelvic space while performing separations, ligations, and suturing, including the challenging vesicourethral anastomosis, with its wristed instruments. The successful procedure, which lasted little more than an hour, fully demonstrated the stability and performance of Carina™.

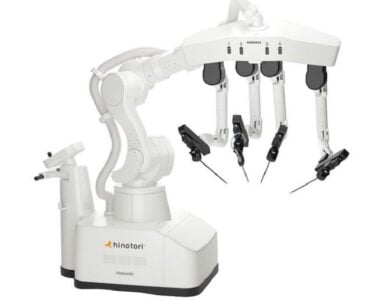
Carina™ in four-arm configuration for first-in-man trial (radical prostatectomy).
Commenting on the completion of the FIM, Prof. Ye said, “the modular architecture poignantly demonstrated the benefits of robotic-assisted surgery (RAS) on Carina™. The surgeon console was simple and efficient, and the instrument arms were dexterous and precise. Both combine to greatly improve the operability of RAS on urologic tumors. This is a major stride forward for a domestic robotic platform.”
Leading up to this major achievement, Carina™ had been used in over 100 animal and cadaver labs for urological, gynecological, colorectal, and thoracic minimally invasive surgeries, including collaborations with KOL surgeons from overseas.
This month, Dr. Eduardo Parra-Davila, one of the world’s leading general surgeons and Director of Minimally Invasive Colorectal Surgery and Hernia/Abdominal Wall Reconstruction at Good Samaritan Medical Center in West Palm Beach (Florida), evaluated Carina™ in a series of animal models and dry labs to see capabilities of the system in the general surgery field, including low anterior resections (LAR), right colectomy, inguinal/ventral hernia repair, bariatric surgery, hiatal hernia repair, and cholecystectomy. He found the modular architecture easily adaptable to his operating workflow, enabling rapid setup for optimal reach and access with a minimal learning curve to use the system – all critical features for abdominal procedures. Additionally, he was able to test the system’s capabilities with challenging surgical maneuvers used during the above procedures that necessitated multi-quadrant access or anterior abdominal wall dissection and suturing.

Dr. Parra-Davila working with Ronovo’s CTO and clinical engineering team during recent on-site hands-on evaluation of Carina™ in multiple animal models and dry labs.
Dr. Parra-Davila pointed out, “First thing that struck me is the easiness to adapt – I can get started on a case with Carina™ in five minutes, and the stability of the system is quite remarkable. I also think Carina™ features many translations from laparoscopy, such as port placement for procedures like LAR and inguinal hernia repair. This is very beneficial for the growing body of laparoscopic surgeons around the world, and makes Carina™ an attractive option in many markets outside of China.”
In June, Dr. Vipul Patel, world-renowned urologic surgeon and Medical Director of AdventHealth Global Robotics Institute, evaluated Carina™ through multiple procedures on live porcine model. The ergonomic design of the surgeon console with high-dexterity hand controls significantly lowered the learning curve and allowed for swift adaptation. Within minutes, Dr. Patel was using Carina’s articulating instruments to perform delicate surgical maneuvers, including dissection, separation, coagulation, and suturing, and easily completed a partial nephrectomy. He was also able to test Carina’s full functionality by successfully simulating a vesicourethral anastomosis.

Dr. Patel engaging with Ronovo’s clinical engineering team members during his on-site hands-on evaluation of Carina™ in multiple animal labs.
Dr. Patel, who had since joined Ronovo’s Clinical Advisory Board, had commented, “I was very impressed, and I didn’t really find that there was a learning curve. I can use the surgical instruments freely, dissect freely, and use energy as needed without issue. And being modular is nice in that you can decide whether you want to use two arms, three arms, four arms, or more. I think that there is definitely utility and merit to that. In my opinion, the system is ready for human clinical trials.”
In April, Dr. Luigi Boni, Professor of Surgery at the University of Milan and Chief of General & Minimally Invasive Surgery at Fondazione IRCCS – Ca’ Granda – Ospedale Maggiore Policlinico di Milano, evaluated Carina™ for multiple colorectal procedures. He successfully performed an LAR with iliac vessel lymph node dissection on a live porcine model, followed by an LAR with total mesorectal excision on a cadaver model with a single docking of Carina. Smooth completion of these procedures exemplified the system’s high level of stability and accuracy as well as ability to perform multi-quadrant surgery.

Dr. Boni successfully completed multiple LARs during his on-site hands-on evaluation of Carina™ in animal and cadaver labs.
Dr. Boni had remarked, “I was pleasantly surprised by the overall performance of Carina™. The high-definition 3D display can visualize the tissue very clearly. The energy platform was particularly easy to use and meets all my needs for LAR. I think Carina™ has unique advantages that will make it very appealing to international markets outside of China as well.”
The successful completion of FIM and these recent KOL surgeon evaluations validate the performance and design intent for the modular architecture of Carina™, which enables versatility to accommodate different surgical workflows for a wide spectrum of endoscopic procedures performed across various surgical specialties.
With this major clinical milestone now in the rear view and a multi-specialty clinical trial planned for later this year, Ronovo is laser-focused on demonstrating the configurability of Carina™ and the adaptability of its instrumentation, which combine to enable the robotic platform to be an agile front-line option for use across urological, gynecologic, colorectal, and thoracic procedures.
Source: Ronovo Surgical