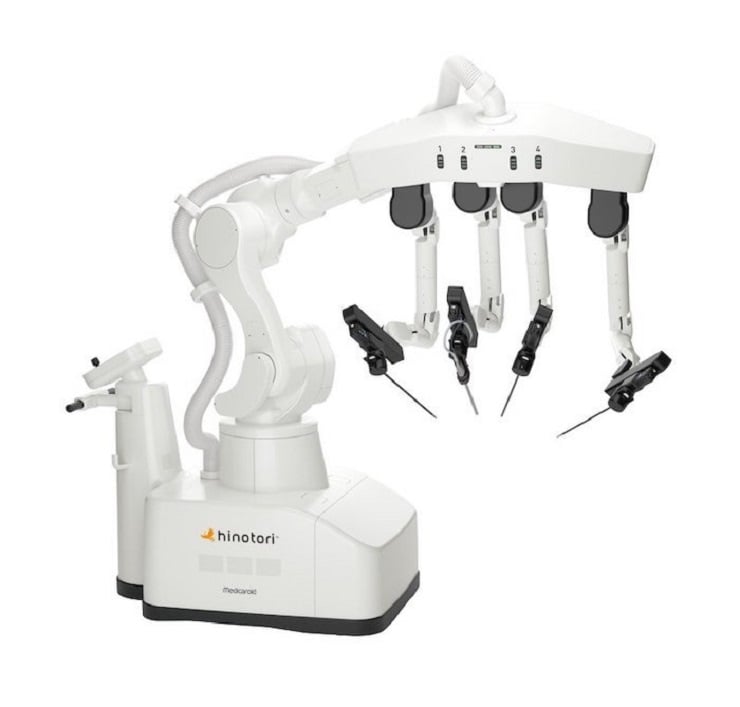
Medicaroid Corporation has announced that it has received the regulatory approval from the Japanese Ministry of Health, Labor and Welfare (MHLW) for the use of the hinotori Surgical Robot System in thoracic surgery.
The hinotori is a made-in-Japan robotic-assisted surgery system which received a Japanese regulatory approval for the use in urology in August 2020, and gastroenterology and gynecology in October 2022. Currently, the hinotori is used in several facilities in Japan. This approval expands the indication for use of the hinotori to include thoracic surgery. The hinotori Surgical Robot System will continue supporting patientfriendly minimally invasive surgery.
Through its medical robots, Medicaroid will continue to support an abundant society where all patients, patients’ family, and healthcare workers can live with peace of mind.
Source: Medicaroid