At the world’s largest medical technology trade fair, KUKA will be showing how robot-based solutions support doctors and therapists.
MEDICA 2023 in Düsseldorf will be the meeting place for the medical technology industry from November 13 to 16. Matching the MEDICA motto “Where healthcare is going”, KUKA will be presenting applications based on the LBR Med that have either already been used successfully in diagnostics or therapy or are still in the approval phase. The spectrum of possible applications ranges from biopsies for brain tumors and ultrasound examinations to rehabilitation after accidents or strokes.

81,000 visitors, around 5,000 exhibitors, 68 percent of them from abroad – these were the figures for the world’s largest medical technology trade fair MEDICA in Düsseldorf in 2022. A strong international exchange is also expected for 2023. The KUKA team is looking forward to welcoming visitors in Hall 10 at Booth A22. Among other things, they can expect to see medical applications based on the seven-axis, sensitive LBR Med, which is the world’s first robot-based component to be certified for integration in medical devices.
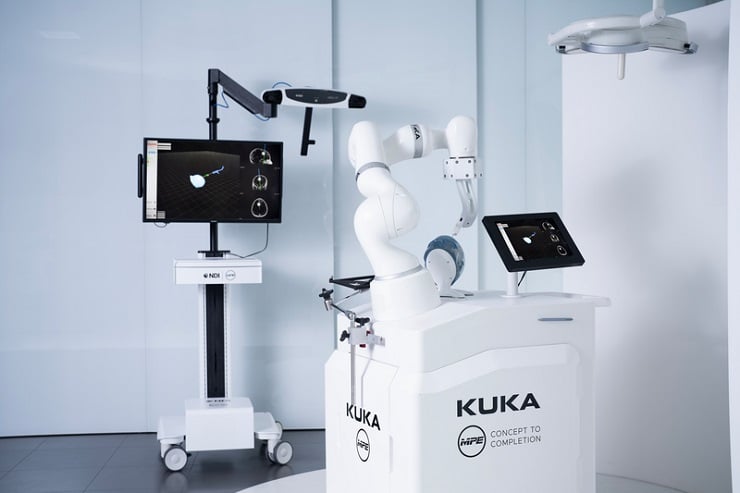
NDI demo cell shows potential application: biopsy for brain tumors
The LBR Med from KUKA was developed specifically for use in medical technology and can be integrated into a wide range of medical technology applications. Its sensitive sensor technology, the comprehensive safety packages, the hygiene-optimized surface and the certification are raising the attention of medical device manufacturers from all over the world. A demo cell at MEDICA shows where the LBR Med could be used after appropriate integration by a medical technology specialist and clinical studies. For example, visitors can see how the LBR Med can contribute to the gentle and safe biopsy of a brain tumor. The biopsy is simulated on a model using real-time tracking based on cameras from the company NDI. With the help of the LBR Med, which is mounted on a cart from MPE, the biopsy needle can be placed. The target and entry positions are planned in advance based on computer tomography data and displayed on an interactive screen.
New features: Movement instead of standstill and more stiffness
“Part of the system is dynamic tracking, which makes it possible for the robot to move precisely with the biopsy needle when the model – or later a patient – moves. The orientation of the needle stays constant, which would prevent the skull wall from being touched or other tissue from being damaged, for example,” explains Alexander Esser, Business Development Manager Medical Robotics at KUKA. KUKA is not planning to launch a medical product on the market, but with the LBR Med it is providing a component for sophisticated medical products. The new “Increased Stiffness” feature, on the other hand, can be understood primarily when trade fair visitors try the robot out for themselves. “As long as the feature is deactivated, the robot is somewhat compliant and yields slightly to touch forces. With the feature activated, the stiffness increases many times over,” explains Dr. Andreas Keibel, Business Development Manager Medical Robotics at KUKA.
Ultrasound examinations non-stop
Another exhibit at the KUKA booth will demonstrate the advantages of haptic telemanipulation with ultrasound. Compared to previous ultrasonic robot cells from KUKA, this one is more user-friendly than ever. Thanks to powerful software, the sensitive LBR Med can be remotely controlled with the transducer. This can significantly reduce the workload of therapists in practices or clinics because they can adopt a comfortable posture during examinations and use a lightweight input device while the robot applies the force for the examinations. While operating the system, the user receives haptic feedback and perceives it when the robot guiding the transducer comes into contact. In addition, workspace boundaries and singularities of the robot are always considered to prevent interference. For ease of operation, the haptic telemanipulation cell can be coupled with a wide variety of operating devices – a seven-dimensional input device from Force Dimension is used at the KUKA booth.
More medical technology options to touch
Because personally tested experiences have more impact than mere observations, visitors to MEDICA can expect a Medical Feature Cell on which they can try out various modes and scenarios for themselves. Andreas Keibel explains: “At this universal demo cell, we demonstrate various performance features of the LBR Med. Hand guiding, teaching by demonstration, force control during trajectory, hand guiding in 6D while maintaining position or orientation, hand guiding along a line or on a given plane, hand-guided zero-space motion – in detail, trade show visitors can try it out for themselves via hands-on.” Whether it’s another ultrasound examination demo, or the example of a robot-assisted spine surgery.
Meet KUKA Partner: Life Science Robotics with rehabilitation robot ROBERT®
An LBR Med user from the very beginning is present at the KUKA booth: Shortly after certification for integration into a medical device according to the CB Scheme, the Danish company Life Science Robotics brought the KUKA Medical Robotics team on board to develop its rehabilitation robot ROBERT®. Until now, this robot has mobilized the lower extremities of bedridden patients who have suffered an accident or stroke, for example. Because it is capable of both passive and active mobilization and, with the aid of sensors, can set up an individual training program, ROBERT® is now in demand worldwide. MEDICA visitors will experience a new feature at the trade fair: ROBERT® now also provides therapy for the upper extremities. The intelligently controlled, active arm therapy with the robot uses sensors to measure the muscle tone of each patient and increase or reduce support as needed.

Flux Robotics: The robotic arm assists the heart surgeon
Flux Robotics, KUKA partner from the Netherlands, will present the Flux One System, a magnet-based technology integrated into a robotic arm of the LBR Med, at MEDICA 2023. This patented navigation enables cardiovascular surgeons and interventional radiologists to maneuver flexible surgical instruments through blood vessels without direct physical contact. This increases the precision and efficiency of surgical procedures. The Flux One system’s magnetically steerable guidewires are specifically designed for use in stenting and other minimally invasive surgery (MIS).
Expansion in the area of high payloads: the new KR QUANTEC HC
Also at MEDICA 2023 there will be a mockup of the new KR QUANTEC HC, which KUKA will launch in 2024. The industrial robot supports a payload of up to 300 kg and can handle heavy medical equipment, such as a linear accelerator used in tumor therapies or an X-ray system in angiography. The new KR QUANTEC HC will have numerous new features such as additional brakes and a personnel recovery system. This allows the brakes to be released manually, so that the KR QUANTEC HC can be moved manually in the case of a power failure, for example.
Picture Captions / Credits:
KUKA@MEDICA_2023_Press (1) / KUKA: With the LBR Med Medical Robotics Feature Demo, trade fair visitors can try out various modes and scenarios for themselves.
KUKA@MEDICA_2023_Press (2) / KUKA: KUKA will present the NDI demo for biopsies with the new “Increased Stiffness” feature at MEDICA 2023.
KUKA@MEDICA_2023_Press (3) / KUKA: Another demo at the show illustrates the advantages of haptic telemanipulation using ultrasound as an example.
KUKA@MEDICA_2023_Press (4) / Life Science Robotics: The rehabilitation robot ROBERT® from Life Science Robotics now also mobilizes patients’ upper extremities.
KUKA@MEDICA_2023_Press (5) / Flux Robotics: The Flux One system from KUKA partner Flux Robotics combines the LBR Med with actively controllable electromagnets for catheter procedures.
Source: KUKA