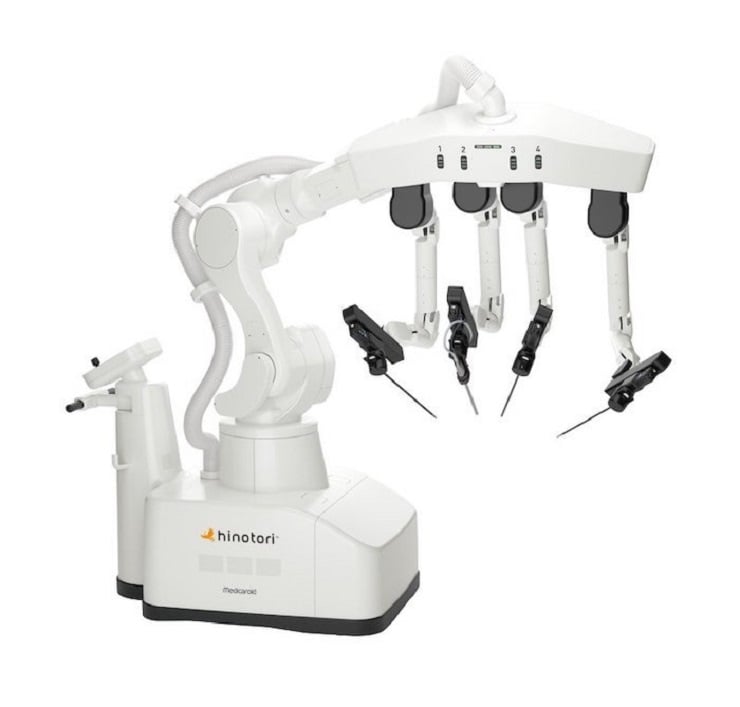
Medicaroid Corporation and Medicaroid Asia Pacific Pte. Ltd. have announced that an application for regulatory approval was filed with the Health Sciences Authority in Singapore for the hinotori Surgical Robot System (hereinafter referred to as “the hinotori”).
In August 2020, Medicaroid obtained a manufacturing and marketing approval for the hinotori from the Ministry of Health, Labor and Welfare (MHLW) in Japan, where it was launched through Sysmex, the general sales agent, in December of the same year. It has since been used in a number of medical institutions and the number of case performed is over 1,000. Along with the domestic business, Medicaroid has been preparing for business expansion in the global market. In December 2022, following the Americas and European regions, a subsidiary in Singapore was established to serve as a base for activities in Southeast Asia, Oceania, and South Asia, with the aim of obtaining regulatory and marketing approval in the region. On April 28, an application for regulatory approval for the hinotori was submitted to the Health Sciences Authority (HSA). This is the first overseas regulatory approval application of the hinotori. An immediate announcement will be made once the approval of the hinotori is obtained.
Through its medical robots, Medicaroid will continue to support an abundant society where all patients, patients’ family, and healthcare workers can live with peace of mind.
Source: Medicaroid