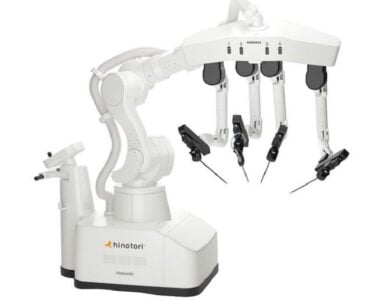
Rob Surgical announces that it has received authorisation to conduct First-In-Human (FIH) clinical trials. This authorisation is issued by the Spanish Agency of Medicines and Medical Devices (AEMPS) and the Ethics Committee of the Hospital Clínic de Barcelona and comes after the success achieved with the preclinical studies, validated and tested in animal models by surgeons from the Mayo Clinic (United States) and the university hospitals Clínic, Vall d’Hebron and Germans Trias de Barcelona. This new milestone brings the medical robotics technology company closer to its founding purpose: to universalise high-precision surgery and bring its benefits to hospitals, surgeons and patients worldwide.
Rob Surgical offers an open and flexible platform consisting of a column of four arms that allow the movements of surgical instruments to be controlled with sub-millimeter precision.
The value proposition is based on three pillars: high precision and safety that benefits the patient, superior usability for the surgeon and a significant reduction in the total cost per surgery that benefits hospitals, as it does not require a dedicated operating room, requires fewer staff, does not require prior training and supports hybrid surgeries.
The differentiating factor lies in the possibility of performing hybrid surgeries, alternating manual and high-precision robot-assisted maneuvers. All this thanks to the use of the same instruments and flexible robotics that allows the robot to intervene only in the most delicate parts, thus reducing the time and cost of each intervention.
The company, created in 2012 as a spin-off of the Polytechnic University of Catalonia (UPC) and the Institute for Bioengineering of Catalonia (IBEC), has raised more than 20 million between private investors and public funding. The founding team is formed by Dr. Josep Amat, Dr. Alícia Casals, Dr. Manel Frigola and the entrepreneur Jaume Amat.
Source: Rob Surgical